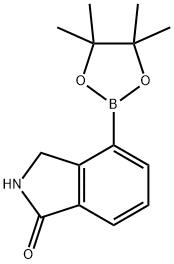
1H-Isoindol-1-one, 2,3-dihydro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-
1H-Isoindol-1-one, 2,3-dihydro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-
CAS: 765948-78-5
Molecular Formula: C14H18BNO3
1H-Isoindol-1-one, 2,3-dihydro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- - Names and Identifiers
1H-Isoindol-1-one, 2,3-dihydro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- - Physico-chemical Properties
| Molecular Formula | C14H18BNO3 |
| Molar Mass | 259.11 |
| Density | 1.16±0.1 g/cm3(Predicted) |
| Melting Point | 189-191 °C(Solv: hexane (110-54-3)) |
| Boling Point | 483.2±45.0 °C(Predicted) |
| pKa | 13.82±0.20(Predicted) |
| Storage Condition | 2-8°C |
| Sensitive | Irritant |
| MDL | MFCD11617913 |
1H-Isoindol-1-one, 2,3-dihydro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- - Introduction
1H-Isoindol-1-one, 2,3-dihydro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-, often abbreviated as TMDO-IK, is an organic compound. The following is a description of its nature, use, formulation and safety information:
Nature:
TMDO-IK is a white to pale yellow solid with the chemical formula C17H23BN2O3S. It has a molecular weight of about 360.26g/mol. TMDO-IK is a hydrophobic compound, almost insoluble in water, but soluble in a variety of organic solvents.
Use:
TMDO-IK are widely used in the field of organic synthesis. As a new type of alcohol protecting group, it can be used in the protection reaction of alcohol. In addition, TMDO-IK can also be used as condensation reagents and catalysts for the synthesis of complex organic molecules, such as natural product synthesis and pharmaceutical research.
Preparation Method:
The preparation method of TMDO-IK is relatively complex and generally requires high chemical experiment technology. A typical synthesis method is to react 1,4-dioxazepane (DMDO-IH) with 4-bromobutanone, and then react with boronic ester under the action of hydrogenation agent to obtain TMDO-IK.
Safety Information:
TMDO-IK compounds are generally relatively stable and safe to use under routine experimental conditions. However, as with other chemical reagents, correct experimental practices and safety measures are still necessary. During use, wear appropriate protective glasses, gloves, etc. At the same time, it is recommended to operate in a well-ventilated area to avoid inhalation or skin contact. During use or storage, care should be taken to avoid ignition and contact with oxidants. If necessary, follow the relevant safety guidelines and operating procedures.
Nature:
TMDO-IK is a white to pale yellow solid with the chemical formula C17H23BN2O3S. It has a molecular weight of about 360.26g/mol. TMDO-IK is a hydrophobic compound, almost insoluble in water, but soluble in a variety of organic solvents.
Use:
TMDO-IK are widely used in the field of organic synthesis. As a new type of alcohol protecting group, it can be used in the protection reaction of alcohol. In addition, TMDO-IK can also be used as condensation reagents and catalysts for the synthesis of complex organic molecules, such as natural product synthesis and pharmaceutical research.
Preparation Method:
The preparation method of TMDO-IK is relatively complex and generally requires high chemical experiment technology. A typical synthesis method is to react 1,4-dioxazepane (DMDO-IH) with 4-bromobutanone, and then react with boronic ester under the action of hydrogenation agent to obtain TMDO-IK.
Safety Information:
TMDO-IK compounds are generally relatively stable and safe to use under routine experimental conditions. However, as with other chemical reagents, correct experimental practices and safety measures are still necessary. During use, wear appropriate protective glasses, gloves, etc. At the same time, it is recommended to operate in a well-ventilated area to avoid inhalation or skin contact. During use or storage, care should be taken to avoid ignition and contact with oxidants. If necessary, follow the relevant safety guidelines and operating procedures.
Last Update:2024-04-09 20:45:29
Supplier List
Spot supply
Product Name: 2,3-DIHYDRO-1H-ISOINDOL-1-ONE-4-BORONIC ACID PINACOL ESTER Visit Supplier Webpage Request for quotationCAS: 765948-78-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,3-DIHYDRO-1H-ISOINDOL-1-ONE-4-BORONIC ACID PINACOL ESTER Request for quotation
CAS: 765948-78-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 765948-78-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: 2,3-DIHYDRO-1H-ISOINDOL-1-ONE-4-BORONIC ACID PINACOL ESTER Visit Supplier Webpage Request for quotationCAS: 765948-78-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,3-DIHYDRO-1H-ISOINDOL-1-ONE-4-BORONIC ACID PINACOL ESTER Request for quotation
CAS: 765948-78-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 765948-78-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
View History

